You've probably had a full fertility workup. Bloodwork, imaging, cycle tracking, maybe a partner's semen analysis. At some point someone asked about your alcohol intake, your caffeine, your weight, your stress. If you're like most patients, no one asked how many hours you actually slept last night. Or the night before that.
Sleep is one of the most underappreciated variables in fertility, a view shared by Stanford urologist Dr. Michael Eisenberg, who has called it “probably the most underappreciated variable in male fertility.” The evidence connecting sleep to reproductive hormones, cycle regularity, sperm quality, and IVF outcomes has been stacking up for years. Why your clinic may not have raised it, what the research actually shows, and what you can do with that information: that's what we’ll cover in this piece.
What sleep is actually doing to your reproductive system
At the level of biology, sleep and fertility share real estate. The hypothalamus, the part of your brain that runs your sleep-wake cycle through the release of melatonin, is also where your reproductive hormones originate. The same control center releases the pulses of gonadotropin-releasing hormone (GnRH) that trigger LH and FSH, the hormones responsible for ovulation in women and testosterone production in men.
Research published in Reproductive Biology and Endocrinology confirms that sleep-generating neurons and reproductive axis neurons occupy the same anatomical location, which means when sleep is chronically disrupted, so is the timing and amplitude of those hormone pulses. You can't knock one off track without dragging the other with it.
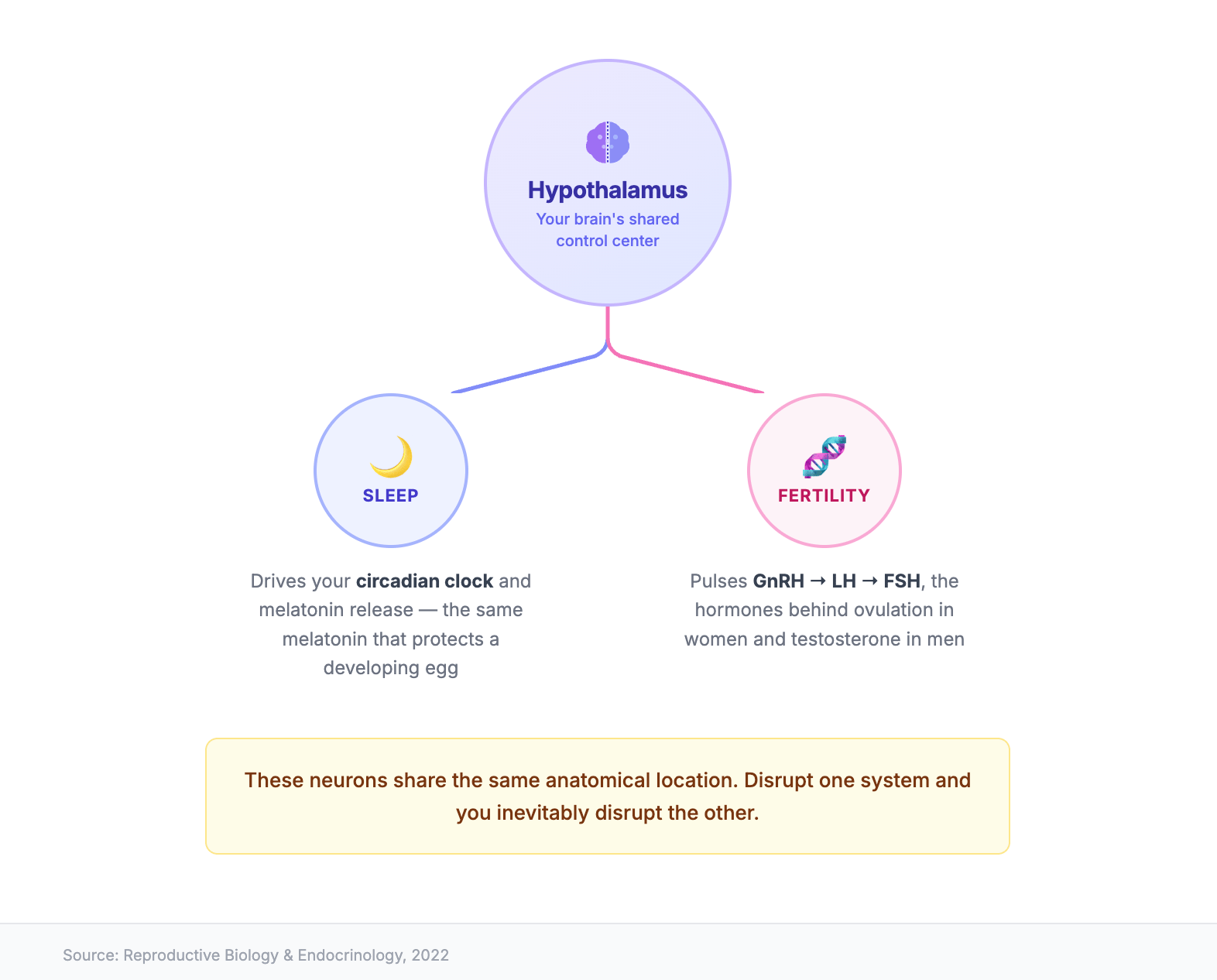
There is also the question of melatonin, which does more than make you drowsy. Johns Hopkins Medicine notes that melatonin is a potent antioxidant, and unlike most antioxidants it is found in concentrated amounts in the follicular fluid that surrounds a developing egg. It appears to protect the egg from oxidative stress during maturation. When you shortchange sleep, you also shortchange the melatonin window that supports that process.
Then there is cortisol, your main stress hormone, which normally peaks in the morning and drops at night. Poor sleep disrupts that rhythm, keeping cortisol elevated when it should be low. Elevated cortisol suppresses GnRH, blunts LH surges, and alters progesterone output. All of which matter for conception.
A 2022 review in Nature Reviews Endocrinology summarized decades of work on this topic with a single line: “disruption of circadian rhythms has a profound effect on reproductive function at every level of the HPG axis.” In other words, it's not that poor sleep might nudge a hormone or two. The system itself is built on the same internal clock that governs when you sleep.
Hear it from a Stanford urologist: The Huberman Lab Podcast: Dr. Michael Eisenberg on male fertility and sexual health Dr. Michael Eisenberg on how sleep shapes testosterone, sperm parameters, and broader male fertility (~2.5 hrs).
What the Data Actually Says About Sleep and Conception
The numbers are specific enough to be useful, though they tell a more nuanced story than most wellness content would have you believe.
The PRESTO study (Pregnancy Study Online), a North American preconception cohort of nearly 7,000 women actively trying to conceive, found that trouble sleeping at night was modestly associated with reduced fecundability (the per-cycle probability of conception). Short sleep duration showed a weaker inverse association: women sleeping fewer than six hours had a fecundability ratio of 0.89 compared to women sleeping eight hours, a signal that didn't reach statistical significance on its own but pointed in a consistent direction.
The male data from the same PRESTO cohort was sharper. Men sleeping fewer than six hours per night had a fecundability ratio of 0.62, meaning roughly 38% lower odds of conception in any given cycle compared with men sleeping eight hours. The relationship was U-shaped: men sleeping nine or more hours also showed reduced fecundability, though that finding was less statistically robust. The practical takeaway is that extremes in either direction appear to matter, especially on the male side.
On the IVF side, a 2022 prospective cohort study of more than 1,200 women undergoing IVF/ICSI found that women sleeping fewer than seven hours per night retrieved 11.5% fewer eggs and 11.9% fewer mature eggs compared with women sleeping seven to eight hours. Women who slept nine to ten hours showed no change in egg numbers but had roughly a 35% decrease in clinical pregnancy rate compared to the seven-to-eight-hour group. Again, the U-shape: not just too little, but too much.
The picture is consistent even where the effect sizes vary. Most of the research points to a practical sweet spot: seven to nine hours, on a reasonably consistent schedule, with relatively few awakenings.
Why Shift Work Gets its Own Chapter
If you are working overnight or rotating shifts while trying to conceive, the research gets sharper and more concerning.
A meta-analysis published in Obstetrics & Gynecology, drawing on data from more than 119,000 women across 15 studies, found that shift workers had a 22% higher rate of menstrual disruption compared with day workers (odds ratio 1.22). The subfertility signal was larger: an odds ratio of 1.80, suggesting shift workers faced roughly 80% higher odds of subfertility in the unadjusted analysis. It's worth noting that after adjusting for confounding factors, the subfertility association weakened and lost statistical significance, which means some of the effect may be explained by other lifestyle variables that tend to accompany shift work (stress, diet, exercise patterns). But the menstrual disruption finding held up, and the proposed mechanism is the same one underneath every sleep-fertility study: circadian disruption and its downstream effects on the hormones that run ovulation and sperm production.
The hardest-hit groups in the literature tend to be nurses, flight attendants, and ER staff: the jobs with both rotating schedules and high physiological stress. Clinicians who specialize in fertility increasingly recognize shift work as a variable worth discussing. Some patients find their OB-GYN or reproductive endocrinologist willing to write a letter supporting a temporary schedule change during active treatment, and the Royal College of Nursing in the UK advises that employers should carry out risk assessments and consider shift modifications when a worker's health is at stake.
This isn't a call to quit your job or to blame yourself for circumstances you can't control. Many people work the shifts they work because that is the work available. But if you are in that situation and you are struggling to conceive, it is worth bringing up with your doctor. A schedule conversation is a legitimate part of a fertility workup, and one that doesn't come up nearly often enough.
The Melatonin Question
Melatonin has become something of a fertility buzzword, and the evidence does support a role. Just not the one supplement marketing tends to suggest.
Your body produces melatonin naturally when it gets dark, and it does far more than make you drowsy. Melatonin is a potent antioxidant that, unlike most antioxidants, is found in concentrated amounts in the follicular fluid that surrounds a developing egg. It appears to protect the egg from oxidative stress during maturation.
In women undergoing IVF, supplementation at around 3 mg per day during the stimulation cycle has been associated with improved mature oocyte yield and higher fertilization rates in some studies. A subgroup analysis found enhanced clinical pregnancy outcomes specifically at doses of 3 mg or less. The proposed mechanism is that antioxidant action in follicular fluid, the same protection your body provides when melatonin production is healthy.
Outside of IVF, the evidence for taking melatonin as a general fertility supplement is thinner. Mayo Clinic cautions that melatonin is still a hormone, one that interacts with other hormones, and that dosing, timing, and long-term use have not been rigorously studied in otherwise healthy adults trying to conceive. At high doses, it may actually interfere with ovulation. Higher is not better. Studies use modest amounts (often 3 mg or less), and there is no evidence that more accelerates anything.
The safer move, if you are interested in the melatonin angle, is to make sure your own body is producing it. That means getting dim light in the evening, avoiding bright overhead light two hours before bed, and giving yourself a genuine sleep window. Your body will make melatonin on its own if you let it.
Sleep Apnea: the Factor Most People Miss
Obstructive sleep apnea (OSA) is one of the clearest sleep-fertility links and also the most under-screened. A systematic review and meta-analysis found that approximately 35% of women with PCOS have obstructive sleep apnea, a striking overlap with the most common endocrine condition in women of reproductive age. A more recent meta-analysis put the pooled prevalence even higher at 37%, with women with PCOS facing more than nine times the odds of OSA compared to women without PCOS. In men, untreated sleep apnea is associated with lower testosterone, higher estradiol, and altered sperm parameters.
If you have been told you snore, wake up gasping, feel unrefreshed after a full night's sleep, or have daytime fatigue that can't be explained by your schedule, that is worth asking your doctor about. A home sleep study has gotten a lot easier to get in the past few years, and treating sleep apnea often improves the exact hormonal profile that matters for fertility.
This applies to both partners. Male-factor issues account for roughly half of fertility difficulties, and sleep apnea is a modifiable male-factor variable that rarely shows up in a standard workup.
What You Can Actually Do
Here is what the research suggests you can actually do, today, while trying to conceive.
Aim for a consistent seven-to-nine-hour sleep window. Consistency (same bed and wake times) matters as much as total duration. Weekend catch-up sleep does not fully compensate for weekday loss.
Keep your bedroom cool and dark. Body-temperature drops are part of sleep onset, and dim evening light preserves your own melatonin rhythm.
Protect the two hours before bed. Limit bright light (especially overhead light and screens) in the wind-down window, and get morning light within an hour of waking. Both tighten your circadian rhythm.
Cap caffeine by early afternoon. Caffeine has a half-life of roughly five to six hours, meaning a 2 p.m. cup is still pharmacologically active at 10 p.m. for most people.
Get honest about alcohol. Even a couple of drinks in the evening fragments sleep architecture, especially REM sleep. If you are trying to conceive, it is worth seeing what a few sober nights do to your sleep quality.
Ask the shift-work question. If you work rotating or night shifts and you are struggling to conceive, talk to your doctor about whether a temporary schedule change is possible. Clinic letters are a real tool.
Screen for sleep apnea. If you snore, wake up gasping, or have daytime fatigue your schedule doesn't explain, ask about a home sleep study. This applies to both partners.
Have the melatonin conversation with your doctor. If you are in an IVF cycle, ask whether your clinic has a view on melatonin supplementation during stimulation. The evidence is modest and dosing matters. This is a doctor conversation, not a supplement-aisle decision.
Look past the sleep score. A wearable's sleep score tracks duration and stages. A tool like OTO Fertility measures whether your nervous system actually recovered, which is a different and more fertility-relevant question.
The Bottom Line
None of this is a promise that an extra hour of sleep will get you pregnant. Sleep is one input in a system with many inputs, some of which you can change and some of which you can't. The reason it is worth taking seriously isn't because it is a secret. It's because it is a foundational variable that rarely gets discussed in a fertility workup, and one that most people haven't fully tried.
Your body is a system, not a checklist. The question isn't "am I doing everything right?" It is "are there modifiable inputs I haven't fully addressed yet?" Sleep, for most people, is one of those.
Resources
Podcasts worth your time: The Huberman Lab with Dr. Michael Eisenberg (Stanford) on sleep, testosterone, and male fertility (~2.5 hrs). The Egg Whisperer Show with Dr. Aimee Eyvazzadeh, which covers sleep among its nine evidence-based fertility risk factors.
Research anchors: PRESTO cohort on female sleep and fecundability. PRESTO cohort on male sleep and fecundability. Fertility and Sterility systematic review on sleep and reproduction. Circadian rhythms within the female HPG axis (Endocrinology, Oxford Academic).
Patient-facing explainers: Mayo Clinic on melatonin. PCOS and sleep apnea meta-analysis. Royal College of Nursing on shift changes.

